 18th July 2008 – 11:00
18th July 2008 – 11:00
Speaker:
Prof. Patricia Targon Campana
Location:
U.M. Grassano, Campus
Abstract:
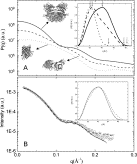
The understanding of the relation between the protein structure and its function is an important challenge in Science, since proteins are involved in all crucial process for life appearing and maintenance. In this sense, the comprehension of stability and conformational mechanisms that can interfere in the protein function is essential to achieve the knowledge and subsequent treatment of several pathologies. Among the most interesting proteins for these studies one can find the lectins: proteins that present the ability to bind carbohydrates specifically and in a reversible way. As carbohydrates can be found in cell membranes, this special feature makes these proteins proper to mediate molecular recognition processes as cell proliferation, cell-cell and cell-virus interactions. Although those proteins have been extensively studied during the last decades, new lectins with interesting properties have been isolated and even the most studied ones presented open questions about their biological function and conformational changes as well. In this sense, this talk aims to present some conformational studies of lectins and other proteins with biotechnological application that have been performed in several structure levels. The changes in secondary structure by means of Far-UV Circular Dichroism, steady-state and phase domain fluorescence at aromatic vicinities and tertiary and quaternary levels using Small-Angle X-Ray Scattering (SAXS) will be presented.


