Thursday, 14th June 2007 – 15:00
Speaker:
Prof. Silvia Morante (Università degli Studi di Roma Tor Vergata)
Location:
U.M. Grassano, Campus
Abstract:
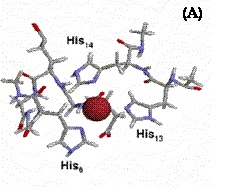
Molecular biology is nowadays strongly overlapped with supramolecular chemistry. An important aspect of supramolecular chemistry, which has recently come to the attention of the scientific community, is the structural role played by metals in intra-molecular and inter-molecular interactions.
Metals are essential elements for many of the fundamental activities of cells and their storing, metabolism and trafficking is mediated by many proteins via well tuned mechanisms because of the toxicity of free ions.
Recently the high sociological impact of neuro-degenerative diseases (like Alzheimer disease, BSE, Parkinson disease, etc.) has pushed the attention of researchers, both from the Biophysical and Biomedical point of view, towards the problem of mis-folding that is a common phenomenon in all amyloidosis pathologies and is regarded as a possible cause of aggregation of protein and plaque formation.
It has been observed that very often plaques contain large amounts of transition metal ions (like Cu+2, Fe+3 and Zn+2). None of the experimental techniques used so far, has been able, however, to unambiguously reconstruct the local structure around the metal ions and elucidate the role they possibly play in the mis-folding process.
I will show the contributions we have given in this direction by using both X-ray Absorption Spectroscopy and ab initio Car-Parrinello Molecular Dynamics in two very much studied systems: Prion Protein and Aβ-amyloids.
Related Information:
Università degli Studi di Roma Tor Vergata


